- होम पेज
- कंपनी प्रोफाइल
-
हमारे उत्पाद
- फार्मास्युटिकल कैप्सूल
- फार्मास्युटिकल इंजेक्शन
- कार्बोप्लैटिन इंजेक्शन आईपी
- रिटक्सिमैब इंजेक्शन आईपी 500 मिलीग्राम
- साइटाराबिन इंजेक्शन बीपी 1g-10ml
- इरिनोटेकन आईपी इंजेक्शन 5 एमजी
- अडालिमुमाब इंजेक्शन
- हेपेटाइटिस बी इम्युनोग्लोबुलिन आईपी
- ट्रास्टुज़ुमाब एम्टैन्सिन इंजेक्शन
- पर्टुज़ुमाब इंजेक्शन
- न्यूकाइन 300mcg इंजेक्शन
- IFOSMA 1000 इंजेक्शन
- नियोस्टिग्माइन इंजेक्शन 0.5mg
- पेगफिलग्रास्टिम इंजेक्शन आईपी
- मैकनेस इंजेक्शन 10000IU
- पैक्लिटैक्सेल इंजेक्शन आईपी
- इंजेक्शन के लिए डेनोसुमाब सॉल्यूशन
- जेमिसिटाबाइन इंजेक्शन आईपी
- फिल्ग्रास्टिम इंजेक्शन 300 एमसीजी
- एंटी स्नेक वेनम इंजेक्शन
- हेपरेल 100 आईयू
- फार्मास्युटिकल टेबलेट
- रिज़ापेन 10mg टैबलेट
- लेडिपसवीर 90mg और सोफोसबुवीर 400mg टैबलेट
- अबीरटेरोन एसीटेट टैबलेट आईपी 500 मिलीग्राम
- पाज़ोपानिब टेबलेट 400mg
- पाज़ोपानिब टेबलेट 400mg
- मेलफालन टैबलेट आईपी 2mg
- वाल्गैनिक्लोविर टेबलेट्स आईपी 450 मिलीग्राम
- एटाग्लोज़िन - 100 टैबलेट
- ओमेगा बी-कॉम्प्लेक्स टेबलेट्स
- ओमेगा एरिथ्रो-250 टैबलेट
- प्रेडनिसोलोन टेबलेट्स आईपी 5 एमजी
- डेक्सामाथासोन टेबलेट्स आईपी 4mg
- पाज़ोपनिब टेबलेट 400mg
- अबीरटेरोन एसीटेट टेबलेट्स आईपी 500 मिलीग्राम
- वेलपनाट टैबलेट्स
- एर्लोटिनिब टेबलेट्स आईपी 150 मिलीग्राम
- संपर्क करें
फिलà¥à¤à¥à¤°à¤¾à¤¸à¥à¤à¤¿à¤® à¤à¤à¤à¥à¤à¥à¤¶à¤¨ 300 à¤à¤®à¤¸à¥à¤à¥
Price 145 आईएनआर/ Unit
MOQ : 100 Units
फिलà¥à¤à¥à¤°à¤¾à¤¸à¥à¤à¤¿à¤® à¤à¤à¤à¥à¤à¥à¤¶à¤¨ 300 à¤à¤®à¤¸à¥à¤à¥ Specification
- पैकेजिंग का प्रकार
- Pre-filled syringe
- खुराक प्रपत्र
- Injection
- मेडिसिन की उत्पत्ति
- India
- लाइफ स्पैन
- 24 months from date of manufacture
- पैकेजिंग (मात्रा प्रति बॉक्स)
- 1 pre-filled syringe per box
- ब्रांड का नाम
- Filgrastim
- नमक की संरचना
- Filgrastim 300 mcg
- संकेत
- Prevention of neutropenia in chemotherapy patients, neutropenia with bone marrow transplantation
- दवा का प्रकार
- सामग्रियां
- Filgrastim 300 mcg
- भौतिक रूप
- फंक्शन
- के लिए सुझाया गया
- Cancer patients undergoing chemotherapy, neutropenia, bone marrow transplantation patients
- खुराक
- As directed by the physician
- खुराक संबंधी दिशा-निर्देश
- Administered subcutaneously or intravenously as prescribed
- के लिए उपयुक्त
- क्वांटिटी
- 1 pre-filled syringe
- स्टोरेज निर्देश
- Store in a refrigerator (2C - 8C). Do not freeze.
- Side Effects
- May include bone pain, headache, fever, skin rash, and nausea
- Route of Administration
- Subcutaneous or intravenous
- Instructions for Use
- Do not shake. Inspect visually for particulate matter and discoloration.
- Appearance
- Clear, colorless to slightly yellow solution
- Shelf Life
- 24 months
- Prescription Required
- Yes
- Strength
- 300 mcg / 0.5 ml
- Regulatory Approval
- Manufactured under WHO-GMP standards
- Container Material
- Sterile glass syringe or vial (as per packaging)
- Contraindications
- Hypersensitivity to filgrastim or other ingredients
फिलà¥à¤à¥à¤°à¤¾à¤¸à¥à¤à¤¿à¤® à¤à¤à¤à¥à¤à¥à¤¶à¤¨ 300 à¤à¤®à¤¸à¥à¤à¥ Trade Information
- Minimum Order Quantity
- 100 Units
- भुगतान की शर्तें
- आपूर्ति की क्षमता
- प्रति महीने
- डिलीवरी का समय
- दिन
- मुख्य घरेलू बाज़ार

Tell us about your requirement

Price: Â
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
मोबाइल number
Email
अधिक Products in फार्मास्युटिकल इंजेक्शन Category
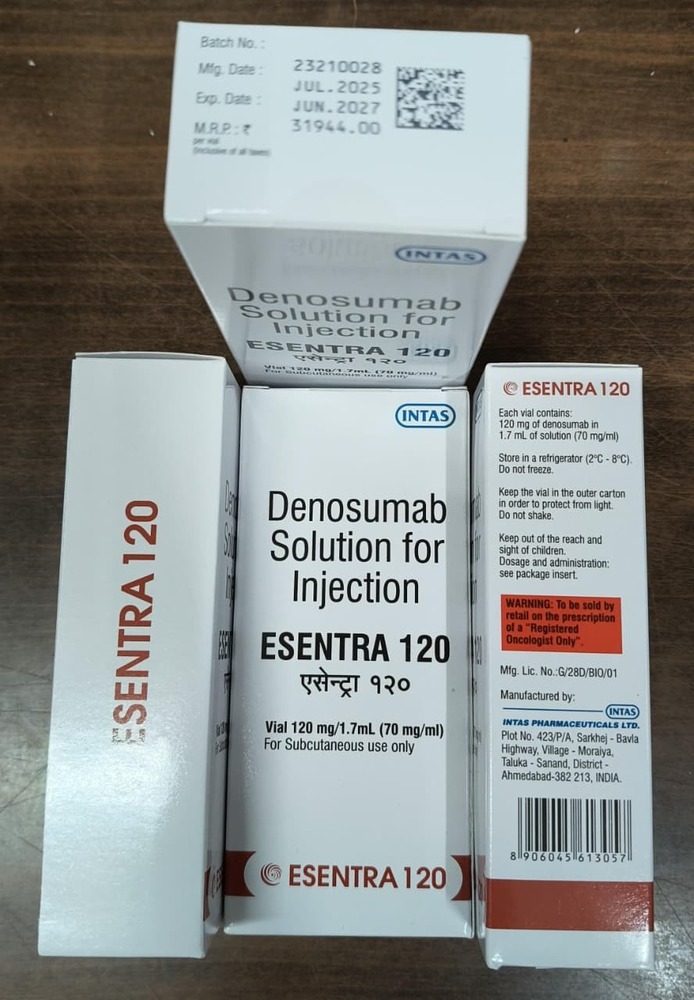
इंजेक्शन के लिए डेनोसुमाब सॉल्यूशन
न्यूनतम आदेश मात्रा : 1
के लिए उपयुक्त : वयस्क
मेडिसिन की उत्पत्ति : भारत
मूल्य की इकाई : पैक/पैक
खुराक : 120 मिग्रा/1.7 मि.ली
माप की इकाई : पैक/पैक
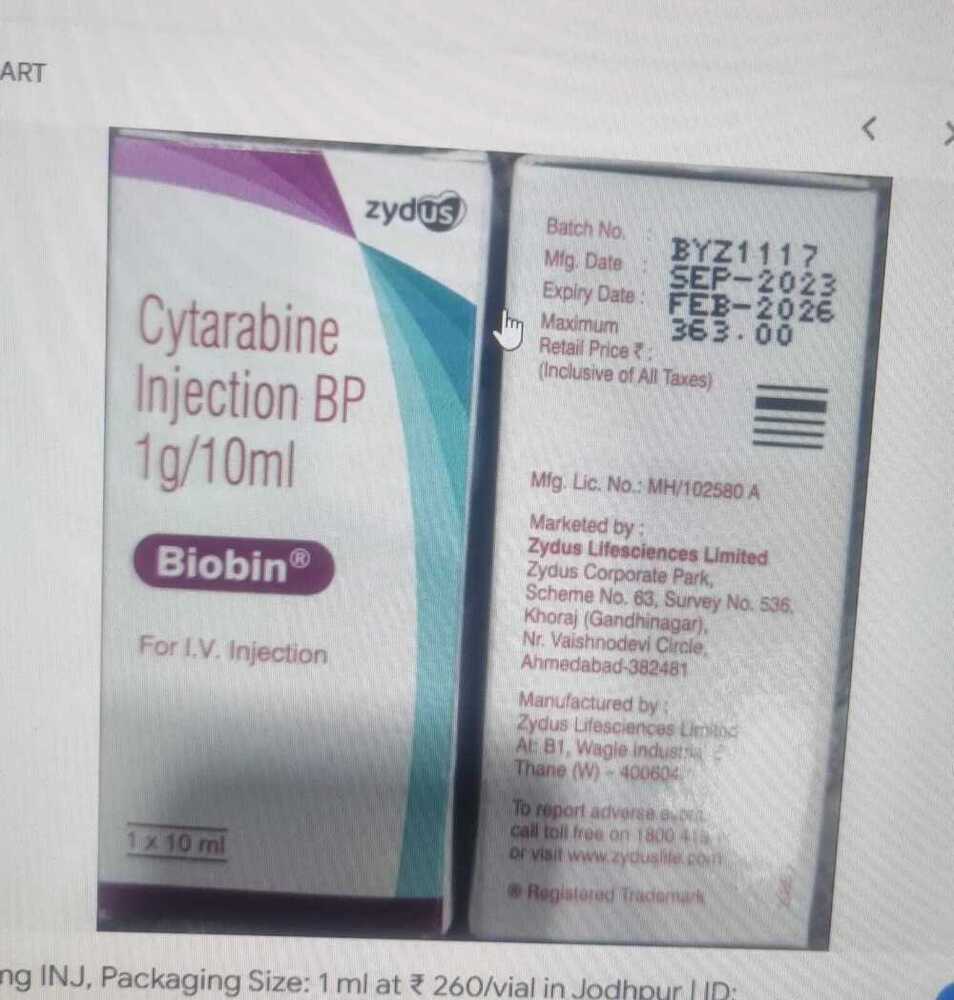
साइटाराबिन इंजेक्शन बीपी 1g-10ml
न्यूनतम आदेश मात्रा : 100
के लिए उपयुक्त : वयस्क
मेडिसिन की उत्पत्ति : भारत
मूल्य की इकाई : पैक/पैक
खुराक : 1 ग्राम10 मि.ली
माप की इकाई : पैक/पैक
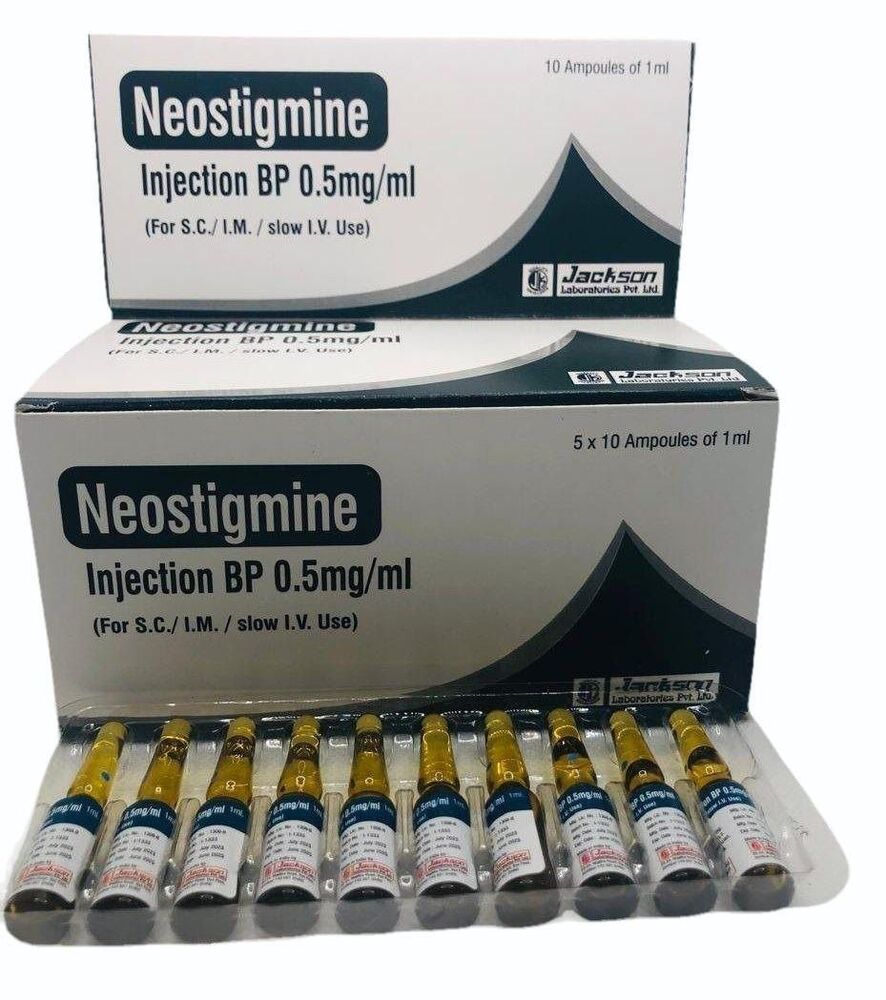
नियोस्टिग्माइन इंजेक्शन 0.5mg
न्यूनतम आदेश मात्रा : 100
के लिए उपयुक्त : वयस्क
मेडिसिन की उत्पत्ति : भारत
मूल्य की इकाई : पैक/पैक
खुराक : 0.5 मि.ग्रा
माप की इकाई : पैक/पैक
पैक्लिटैक्सेल इंजेक्शन आईपी
न्यूनतम आदेश मात्रा : 1
के लिए उपयुक्त : वयस्क
मेडिसिन की उत्पत्ति : भारत
मूल्य की इकाई : यूनिट/यूनिट
खुराक : 300 मिलीग्राम / 50 मिली
माप की इकाई : यूनिट/यूनिट


 जांच भेजें
जांच भेजें

 मुझे निःशुल्क कॉल करें
मुझे निःशुल्क कॉल करें
 English
English Spanish
Spanish French
French German
German Italian
Italian Chinese (Simplified)
Chinese (Simplified) Japanese
Japanese Korean
Korean Arabic
Arabic Portuguese
Portuguese